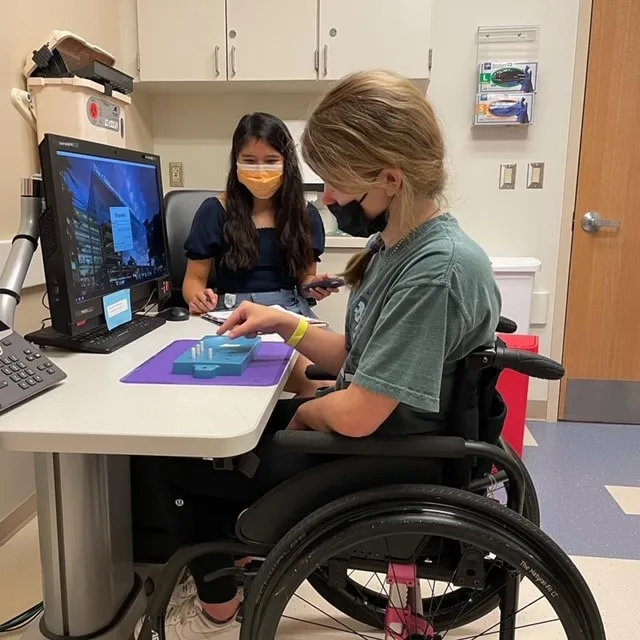
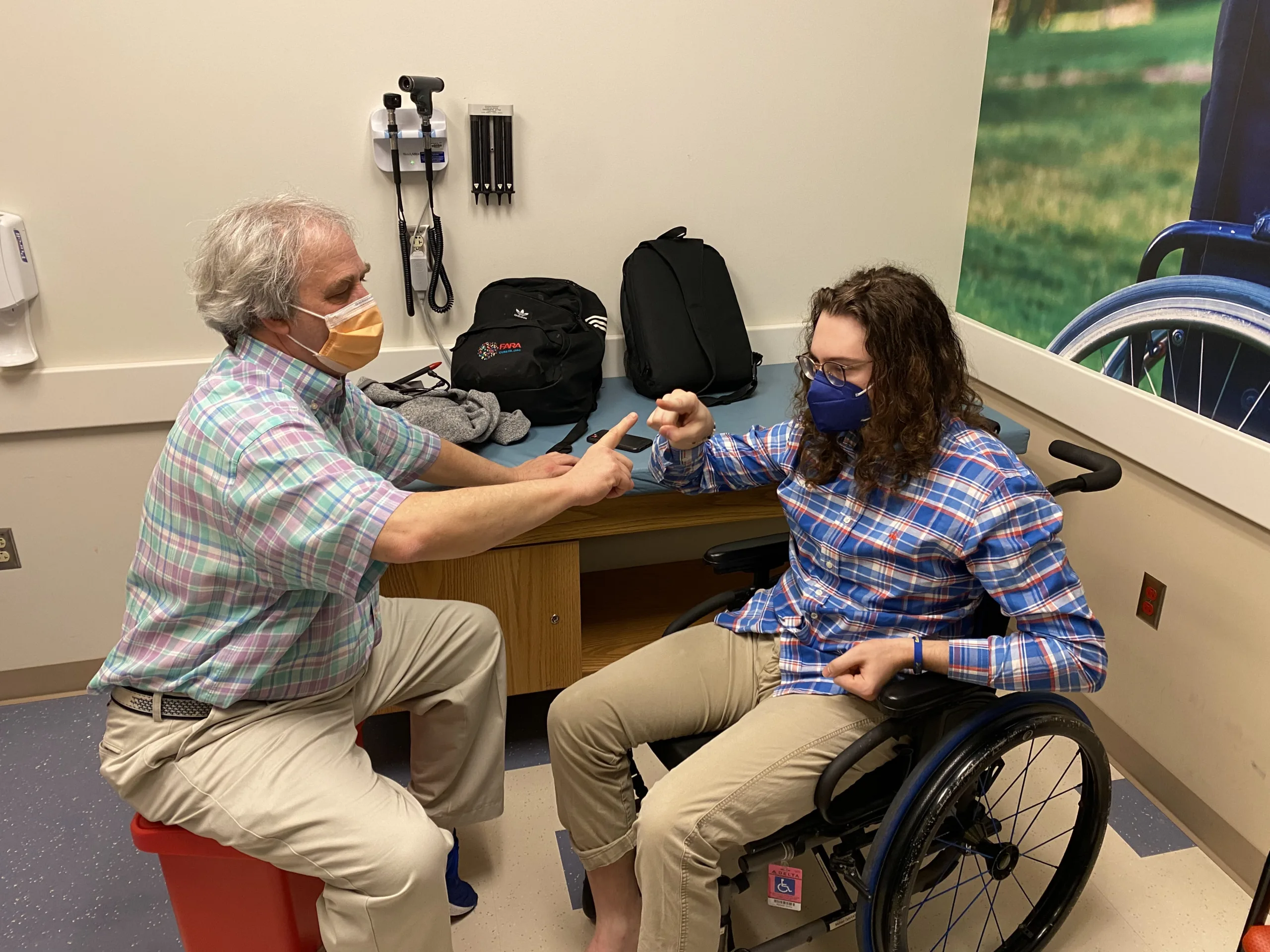
Sites are led by investigators who are experts in FA and staffed by clinical trial ready study teams. At each clinical research center there is a team of researchers, physicians and health care providers dedicated to FA.
A GLOBAL RESEARCH NETWORK
Friedreich’s Ataxia Global Clinical Consortium
The Friedreich’s Ataxia Global Clinical Consortium (FA GCC) is an international group of clinical research centers that work together to advance treatments and clinical care for individuals with Friedreich’s ataxia. The consortium collaborates with pharmaceutical companies, government agencies, other research centers, and the patient community to facilitate clinical research and trials needed to identify new therapies.
FA GCC Site Locations
Prof. Dr. med. Kathrin Reetz
Prof. Dr. Sinead Murphy
Prof. Dr. med. Thomas Klopstock
Mohammed Faruq
FA GCC sites provide infrastructure to conduct studies and trials in Friedreich’s ataxia.
Prof. Dr. med. Kathrin Reetz
Prof. Dr. Sinead Murphy
Prof. Dr. med. Thomas Klopstock
Mohammed Faruq
UNIFAI Natural History Study
A single global natural history study protocol was developed by the FA GCC, called the UNIFAI Study. The UNIFAI Study collects natural history data and improves the clinical outcome assessments that can be used in FA clinical trials.
For more information, please visit UNIFAI or clinicaltrials.gov.
The FA GCC enables collaborative global clinical research, accelerates the development of new therapies, and improves outcomes for those living with FA.
Anyone considering participating in a clinical trial should discuss the matter with their physician. FARA does not endorse or recommend any particular studies.
Unifying the Global FA Community
For Individuals with FA and Families
Each of the FA GCC sites is a place where people with FA can go to participate in research studies, meet with expert physicians and healthcare providers knowledgeable in FA and in many cases receive clinical care. At every site researchers are conducting natural history, clinical outcome assessment and biomarker studies. These are studies that document the clinical symptoms and progression of FA and evaluate specific tests or measures, such as a neurological scale, timed walk, and quality of life measures, that can be used in future clinical trials. All of the research data collected at each site is gathered into a combined database. Some sites have additional studies, such as vision evaluations, speech recordings, motion sensor collection, balance tests, biomarker studies, and clinical trials.
The investigators and coordinators at each of these sites are very familiar with medical issues related to FA and can provide clinical services as well. You can contact the sites to learn about the studies and clinical services being offered.
For Academic and Industry Researchers
- Planning, implementation and recruitment of clinical trials – The FA GCC investigators and site staff are experienced in the planning and conduct of clinical trials for FA and other neurodegenerative diseases. Services utilized can range from planning consultation to full implementation and coordination of a study.
- Promoting understanding of natural history and development of clinical outcome assessments to inform trial design – The FA GCC was formed from a group of investigators who were previously collaborating to develop clinical outcome measures in FA. Longitudinal data is available and continues to be collected and shared publicly with the FA research community. A list of such publications is accessible here. Click here to connect to the Friedreich’s Ataxia Integrated Clinical Database (FA-ICD).
- Conducting clinical research and trials – Partnership for new clinical studies to support the advancement of novel interventions, such as biomarker development, new outcome measure development, and genetic studies. The FA GCC can function as the clinical site for a new study, providing access to patients and their relevant clinical data, handling the procurement of biological tissues and fluids, and assisting with the preparation and filing of the necessary regulatory approvals.
Researchers who would like to learn more about the FA GCC or discuss a study should contact the FA GCC Co-chairs:
- Dr. Med. Jörg B. Schulz AtaxieStudien@ukaachen.de
- David Lynch, MD, PhD, FAPROGRAM@chop.edu, +1-215-590-2242
- or FA GCC Coordinator: fagcc-info@curefa.org
Partnering with Industry
The investigators and coordinators in the FA Global Clinical Consortium are incredibly responsive and professional. With their partnership and FARA leadership, we were able to identify interested investigators and sites, develop a phase 2/3 protocol, obtain regulatory and ethics committee approvals, and launch a study in approximately six months. Once the study opened for screening, the first cohort of patients was identified, screened and enrolled in less than two months.
–Colin J. Meyer, PhD, Former Vice President Product Development & Chief Medical Officer, Reata Pharmaceuticals
For Physicians
The FA GCC can assist you in offering your patients access to clinical research studies in FA. The investigators and coordinators at each of the sites can work with you to provide guidance on updates in clinical care and management for your patients.
Core Objectives
- Unifying the global FA community (clinician researchers and patients) contributing to the understanding of disease natural history.
- Creating a secure and sustainable data platform to accelerate the implementation of innovative clinical studies and facilitate interventional therapy development.
- Optimizing and expanding the clinical outcome assessments and biomarkers in FA that are necessary for clinical trials.
- Tracking health problems and progression of symptoms that occur throughout the disease course and evaluating changes in natural history as new disease-specific therapies are approved.
- Facilitate genetic modifier and biomarker studies.
- Create a global mechanism for sharing data and resources with basic researchers and clinical investigators.
- Generating a registry of individuals with Friedreich’s ataxia to aid recruitment to future trials and studies.
- Contribute to the evolution of clinical practice guidelines, informing clinical care and improving outcomes for people with FA.
FA GCC Resources
Clinical Data Collection Worksheets & Assessments
There are a number of clinical research tools available for download such as data collection worksheets for mFARS, Functional Disability Staging, and FA Activities of Daily Living.
FA GCC Project Proposal Form
If you have a project proposal where you’d like to request use of FA GCC resources, please download, complete, and send this form to the FA GCC Coordinator at fagcc-info@curefa.org.
FA GCC Unified Natural History Study (UNIFAI) - Update December 2025
In our first newsletter, we share key highlights from the cohort so far, including the countries contributing data, the number of visits completed, how the cohort is distributed across disease stages and ages, and insights into the cohort’s broader clinical profile.
Structure and History
The FA GCC is comprised of a funding board, committees, working groups, a patient advocacy/advisory team and the investigator members of each clinical site.
The Scientific Steering Committee (SSC) provides scientific leadership with respect to the FA GCC Research Activities. The representatives serving on the SSC will include, but may not be limited to:
- The Co-Chairs – assume overall responsibility for the proper functioning of the SSC and will chair the SSC meetings. They will also act as spokespersons about the Research Activities to the public.
- Up to eight Investigators from Research Sites based on regional representation.
- One or more representatives from each of FARA and FARA Europe, the Patient Advisory Team, and the Management Team.
FA GCC Scientific Steering Committee
Co-Chairs
Prof. Dr. Jörg Schulz (DE) & Dr. David Lynch (US)
Investigator members
Prof. Dr. Massimo Pandolfo (CA)
Dr. Kathy Mathews (US)
Dr. Louise Corben (AUS)
Dr. Caterina Mariotti (IT)
Prof. Paola Giunti (UK)
Board / Management Team / PAT
Jennifer Farmer (FARA)
Myriam Rai (FARA Europe)
To be appointed (Patient Advisory Team)
Workgroups can be proposed by Research Sites, the SSC or Management Team, as needed to carry out supporting activities related to Research Activities and FA GCC related interests (e.g., evaluating future research areas or technologies). Workgroup participants engage in a collaborative manner with each other on a specific project or research question/needs for which the Workgroup has been established. Workgroup meetings will be conducted on an as needed basis but no less than quarterly.
The success of FARA’s first network of clinical research centers, the Clinical Research Network in FA (CCRN in FA), has allowed for further expansion globally and harmonization with The European Friedreich’s Ataxia Consortium for Translational Studies (EFACTS), resulting in one harmonized Consortium, the Friedreich’s Ataxia Global Clinical Consortium (FA GCC).
The FA GCC is built on the expansion of the CCRN and coming together of two existing natural history studies:
- FA-COMS, supported by FARA, conducted in the US, Canada, Australia, New Zealand and India and;
- EFACTS, a natural history study conducted throughout Europe.
These studies have been carried out in parallel for over a decade, with many similarities. The FA GCC has combined the existing networks, leveraging current resources and staff. This new structure encourages balanced global interaction and collaboration.