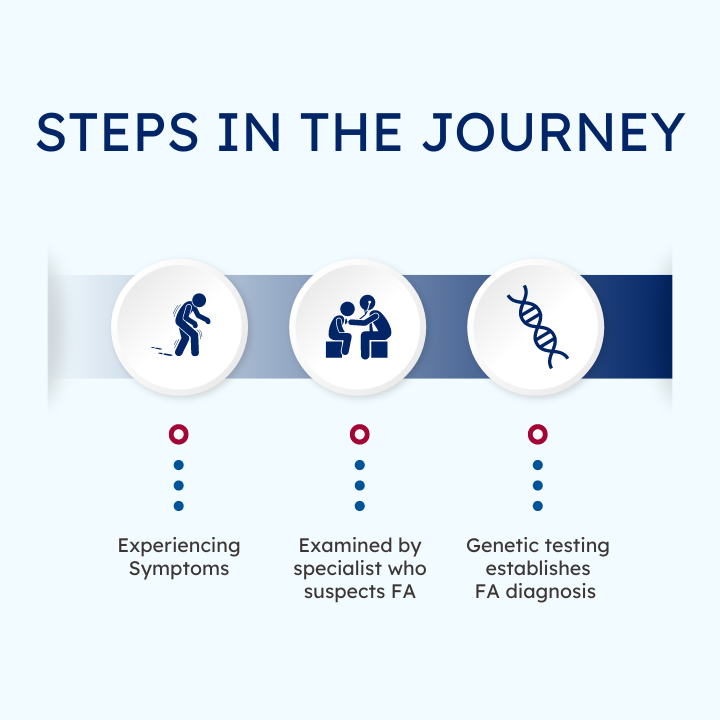
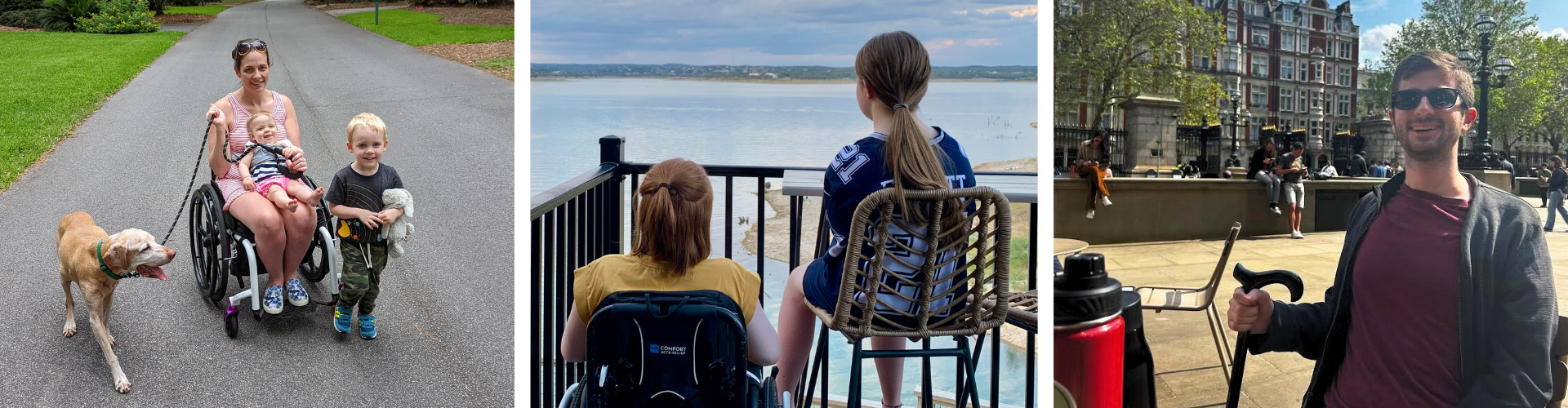Journey to an FA Diagnosis
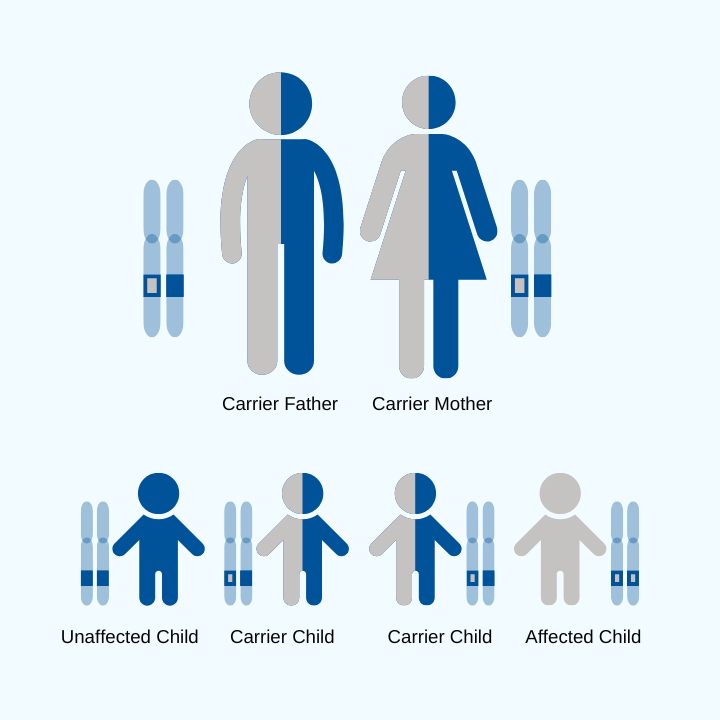
FA is a genetic disease, which means genetic testing is the gold standard for diagnosis. The genetic test will examine the causative gene in FA, FXN.
Most people begin experiencing symptoms and receive a diagnosis between the ages of 5 and 15 years. About 25% of people have late onset FA, and they begin experiencing symptoms in adulthood.
Each person with FA has a different journey that leads them to a genetic diagnosis. Click below to read more.